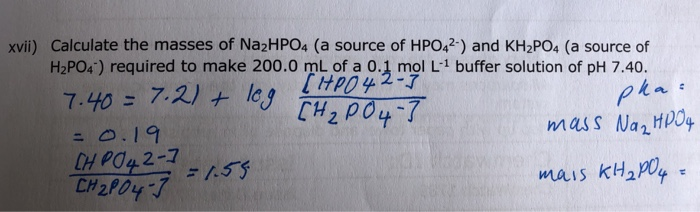The percentage composition of sodium phosphate as determined by analysis is 42.1% sodium, 18.9% phosphorus and 39% oxygen. Find the empirical formula of the compound (work to two decimal places). (R.A.M: Na = 23, P = 31, O = 16.)
How to calculate the pH of a buffer solution obtained by dissolving 12.0 g of KH2PO4(s) and 27.0 G of Na2HPO4(S) in water and then diluting to 1.00 L - Quora

Aqueous solutions of NMA, Na2HPO4, and NaH2PO4 as models for interaction studies in phosphate–protein systems - ScienceDirect

Comparative Study of Sodium Phosphate and Sodium Sulfate in Aqueous Solutions at (298.15 to 353.15) K | Journal of Chemical & Engineering Data


![Glycine [C2H5NO2] Molecular Weight Calculation - Laboratory Notes Glycine [C2H5NO2] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/06/glycine-molecular-weight-calculation.jpg)



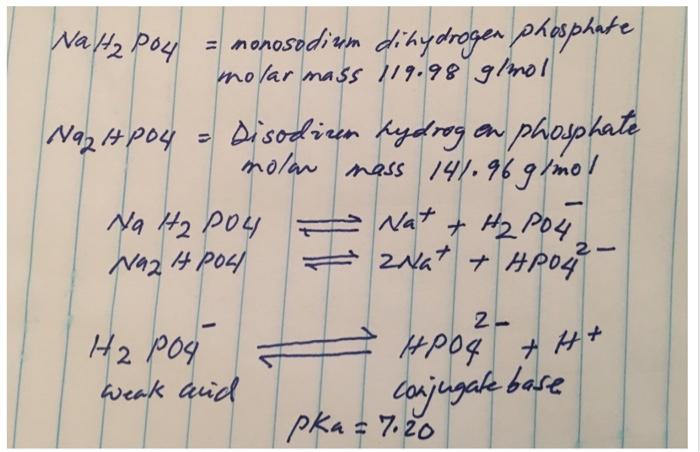

![Answered: [A-] pH= pka + log [HA] a. Determine… | bartleby Answered: [A-] pH= pka + log [HA] a. Determine… | bartleby](https://content.bartleby.com/qna-images/question/0efbec1c-3aeb-47c8-90fa-39b6de84bb74/c728bc90-2d81-4d3c-b78c-7dd9d14854f6/k04zzb.jpeg)