Catalytic C–H Bond Activation and Knoevenagel Condensation Using Pyridine-2,3-Dicarboxylate-Based Metal–Organic Frameworks | ACS Omega

The plausible mechanism of the Knoevenagel condensation in the medium... | Download Scientific Diagram

Enzymatic Tandem Approach to Knoevenagel Condensation of Acetaldehyde with Acidic Methylene Compounds in Organic Media - Koszelewski - 2017 - European Journal of Organic Chemistry - Wiley Online Library

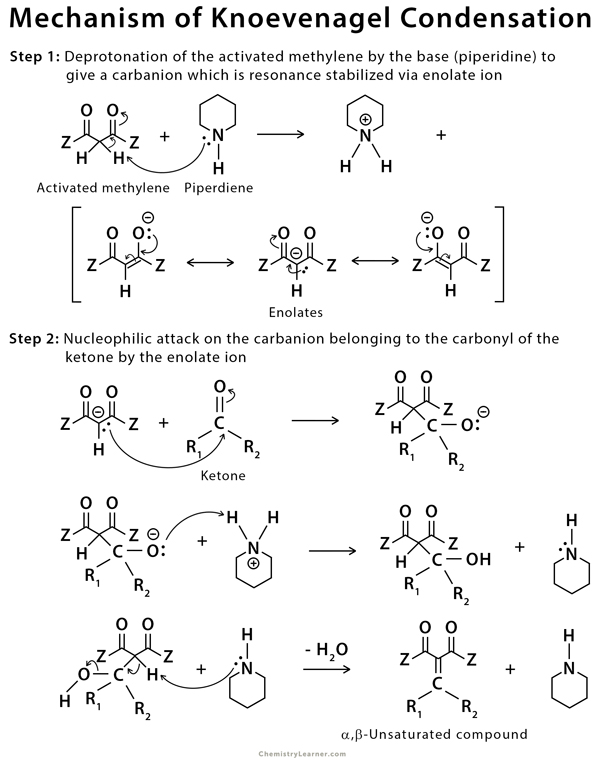
The Knoevenagel reaction is a carbonyl condensation reaction of an ester with an aldehyde or ketone to yield an \alpha , \beta -unsaturated product. Show the mechanism for the Knoevenagel reaction

Kinetics Studies on a Multicomponent Knoevenagel−Michael Domino Reaction by an Automated Flow Reactor - Haas - 2019 - ChemistryOpen - Wiley Online Library

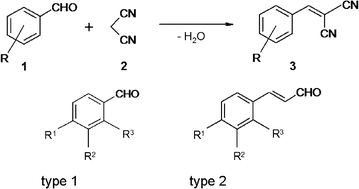
Base-catalyzed Knoevenagel condensation reaction (synthesis of compound... | Download Scientific Diagram