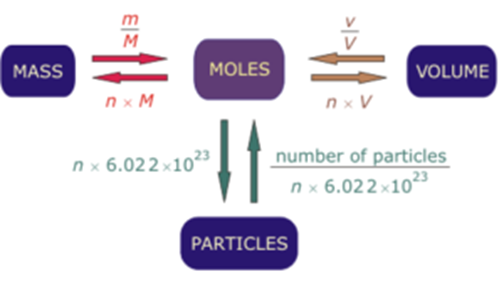
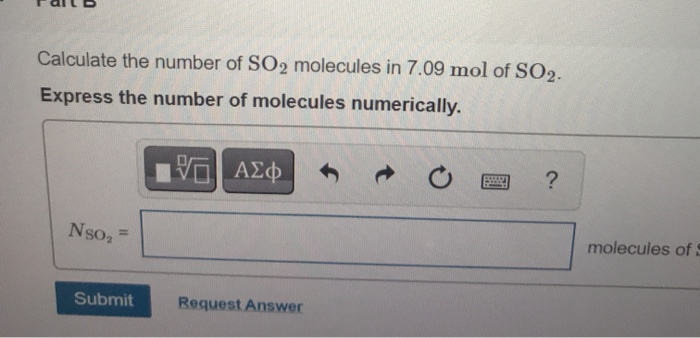
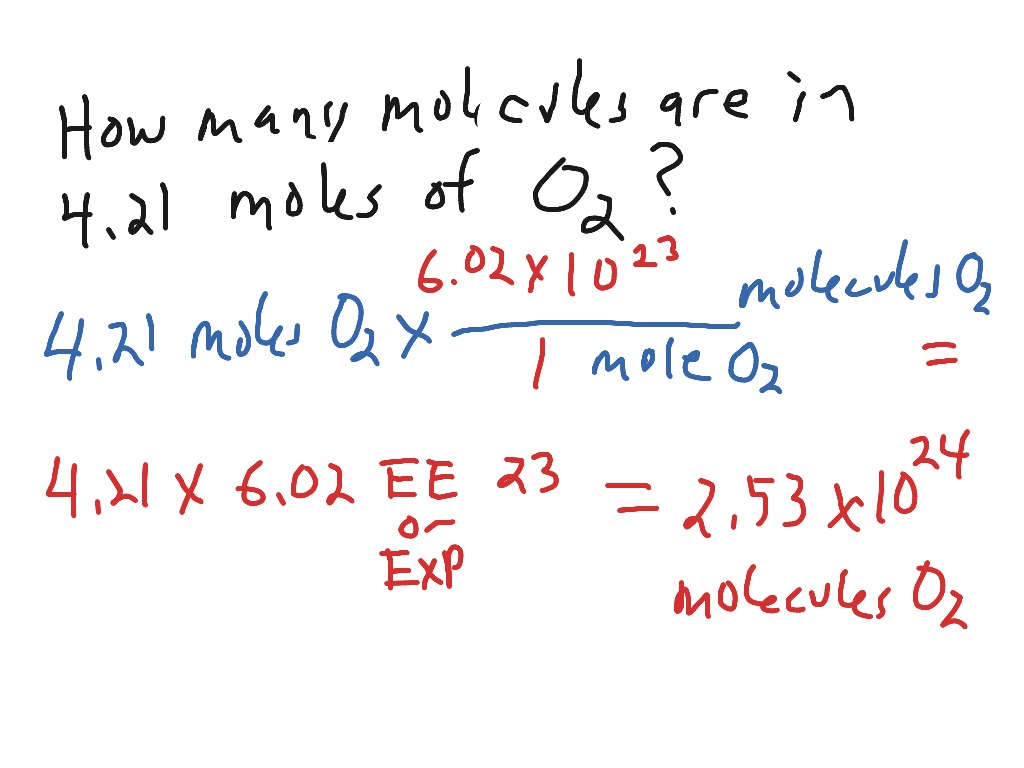Calculate the number of moles and the number of molecules present in 1.4 g of ethylene gas. What is the volume occupied by the same amount of ethylene?A. 1.25 litresB. 1.12 litresC.

Calculate the number of molecules in 2 xx 10^(-6)m^(3) of a perfect gas at 27^(@) C and at a pressure of 0.01 mm of mercury. Mean KE of a molecules at 27^(@)
How to calculate the number of molecules of an element in a compound if I only know the total mass - Quora